Fourteen years ago, at age 75, my father had his first serious heart attack. During an event like that, heart tissue is starved of oxygen and can die. Tests showed my dad’s heart had been badly compromised.
It wasn’t clear what to do. Over his five weeks in hospital, the cardiologists swayed back and forth among options. Should he get a “redo” – a second bypass surgery, grafting new arteries to feed the heart, to follow up on the quadruple bypass he’d had 12 years earlier? Or angioplasty to widen the arteries, with stents to keep them propped open? Or was it safest just to treat him with drugs alone? In the end, he got angioplasty and multiple stents, followed by a diet of pharmaceuticals.
It obviously worked – he’s still alive and kicking – but I remember wishing in the midst of the crisis that he had a stash of stem cells, to regenerate his heart from within. He didn’t. But now Paul Santerre, a U of T biomedical engineering professor leading a research team at the Ted Rogers Centre for Heart Research, is at work on the next-best thing – an injectable polymer material studded with cardiac cells that can take up residence at the site of injury. He calls it the cardiac tissue patch.
Bypass and angioplasty and stents and medicines can all work fine, but they are workarounds. Being able to regenerate cardiac tissue, as Santerre is aiming to do, will allow the heart to function closer to the way it did before the injury.
One of the big challenges of crafting synthetic materials that will be put inside the human body is that our immune system is expert at sniffing out foreign substances – then attacking them, breaking them down and tossing them out. This is terrific if the foreign substance is a pathogen, such as measles or E. coli, but not so good when it’s a medical device.
Santerre has spent years pondering this problem. He knows that the immune system’s response to foreign objects is a lot like its response to a wound. The first thing that happens is inflammation, during which damaged (or foreign) cells are removed. This is the downfall of many a medical implant. After that, however, wound-healing begins – the laying down of new blood vessels and the groundwork for new tissue – and that rebuilding phase was something Santerre was keen to harness for heart regeneration.
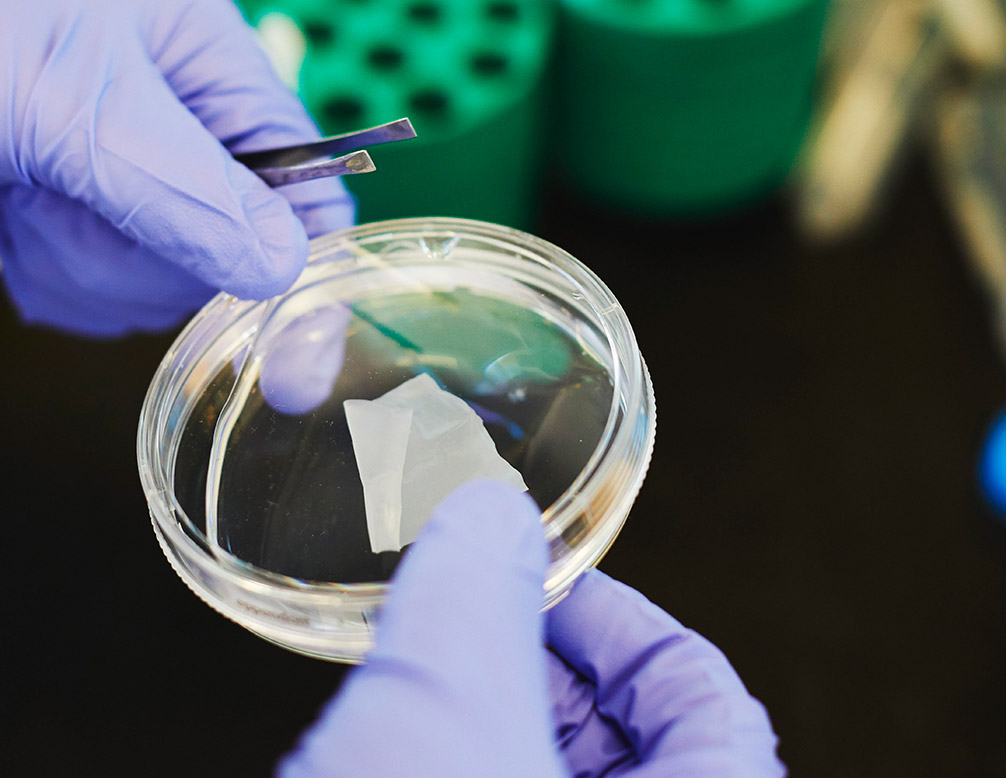
He was particularly interested in creating a material that could act as a scaffold for cardiac muscle cells. These cardiac cells, which had been derived from stem cells, were extremely promising, but when they were injected into the heart on their own, they ended up disorganized. Out of millions of cells injected, fewer than 10 per cent typically survived.
Santerre’s fine polymer is designed in such a way that it helps orient the cardiac cells, so that they fall into their required pattern. This orientation is very important for heart muscle function – it ensures that the receptors on the cells align correctly, so that the cells together can send the electrical pulses needed to keep the heart beating. “If they are oriented in every which way,” says Santerre, “then the force doesn’t really go anywhere.”
The polymer is designed to chemically mimic a natural protein, so it doesn’t arouse suspicion from the immune system. “What you do is you enable the local proteins to interact with this polymer as if it belongs there,” says Santerre. His experiments show that white blood cells that would normally attack foreign substances like these don’t pay much attention to his. Not only does this mean that the immune system is less likely to attack, it also means that the patch might be incorporated into the attempts at wound-healing that follow.
The idea of the patch, then, is to use the sheet of oriented cells in a damaged part of the heart, and coach the cells to develop into functioning heart muscle tissue.
The polymer, which looks like fine white gossamer, is so thin it could be folded and drawn up into the heart through a catheter. Chopped into tiny pieces, it could be injected straight into the heart with a needle. The polymer is also designed to degrade well, says Santerre. After it helps regenerate healthy tissue, it has no purpose in the body. Eventually, water will break the material down into carbon dioxide and a naturally digestible product, which can be excreted.
The patch won’t be ready in time for someone of my dad’s generation. It’s still a few years away from being tried out in a mouse. Santerre says he and his colleagues will spend the next year or so perfecting the sheet of cardiac cells. Then they have to see if the cells can retain their appearance and behaviour in the lab. “If they hold on to these things in my petri dish, they’re going to be able to hold on to them in their appropriate ecosystem,” says Santerre. Be still my beating heart.
In 2014, the Rogers family donated $130 million to U of T, the Hospital for Sick Children and the University Health Network to create the Ted Rogers Centre for Heart Research. Scientists and clinicians at the centre, such as Paul Santerre, are working to reduce hospitalizations due to heart failure and improve the lives of people living with heart disease.
Recent Posts
For Greener Buildings, We Need to Rethink How We Construct Them
To meet its pledge to be carbon neutral by 2050, Canada needs to cut emissions from the construction industry. Architecture prof Kelly Doran has ideas
U of T’s 197th Birthday Quiz
Test your knowledge of all things U of T in honour of the university’s 197th anniversary on March 15!
Are Cold Plunges Good for You?
Research suggests they are, in three ways





