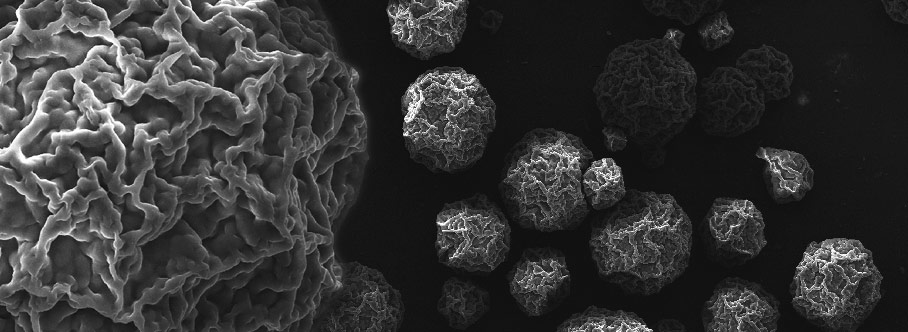
Ever since 1961, when U of T scientists published a landmark paper about the potential of stem cells, researchers in regenerative medicine have been trying to figure out how to use these cells to replace damaged tissue or organs.
Michael Sefton, the executive director of U of T’s Medicine by Design, focuses on new treatments for diabetes. People with diabetes can’t produce the insulin they need to regulate their blood sugar levels. Left untreated, the condition causes nerve and vascular damage, and raises the risk of heart disease, stroke and even blindness.
Sefton, a University Professor in chemical engineering and at the Institute of Biomaterials and Biomedical Engineering, is developing a technique that could someday treat diabetes with a single injection. He is creating tiny capsules that could be injected into a patient with diabetes, where they would deliver special pancreatic cells capable of restoring proper insulin function.
As Sefton explains, the capsules, made of collagen, are a few hundred microns in size – small enough that they can pass through a syringe needle. Each one contains clusters of about 3,000 pancreatic cells called “islets” that regulate insulin production. Sefton says the cells will eventually be engineered to prevent the patient’s body from rejecting them.
A challenge for Sefton and his research team is that people with diabetes suffer damage to their blood vessels, which makes it nearly impossible for the islets to do their intended job. Think of transport trucks (the capsules) trying to drive on an unpaved road (the damaged blood vessels). The capsules also have to be engineered to trigger the growth of healthy new blood vessels – akin to the transport trucks generating a freshly paved highway for themselves.
By the time the research is complete, the islets will be “retooled to maximize their performance,” Sefton says, allowing them to survive the initial and most difficult phase of their journey into the body.
So far, Sefton and his team have been refining the process on diabetic mice. With funding from the Juvenile Diabetes Research Foundation, the next phases – which will take about two years – involve perfecting this technique for rats and then pigs. “It should work in rats but it’s not clear it will scale to pigs,” he says. With larger animals, the therapy requires hundreds of times more injectable cell-containing modules, as well as methods that ensure these tiny “trucks” are deployed properly in the patient’s body.
As Sefton points out, regenerative medicine has made great advances, but the tissues that have seen the greatest success – such as joints and bones – tend to be less complex, with fewer cells. The islet modules, though still in early development, point to the next chapter in regenerative medicine, which will target more complex types of organs and tissues. It’s a future that could even see cellular machines zipping through our veins, helping to fix stuff.
Recent Posts
People Worry That AI Will Replace Workers. But It Could Make Some More Productive
These scholars say artificial intelligence could help reduce income inequality
A Sentinel for Global Health
AI is promising a better – and faster – way to monitor the world for emerging medical threats
The Age of Deception
AI is generating a disinformation arms race. The window to stop it may be closing