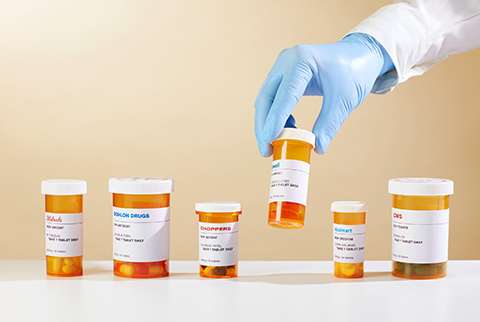
When Toronto researcher and family doctor Nav Persaud found no scientific evidence that the common morning-sickness drug Diclectin, widely prescribed in Canada over the past 40 years, is effective, he had a startling realization. “I began to wonder, are there other common medications that might be ineffective also?” Persaud, a professor in U of T’s department of family and community medicine and a staff physician and scientist at St. Michael’s Hospital, headed up a small team of researchers to find out.
They began by drawing up a list of so-called essential medicines. Back in 1977, the World Health Organization (WHO) created this concept, and today each of at least 130 countries has its own list. Compiling a master file from these was a Herculean task, since the lists have different medicines considered essential (tropical medicines, for example, appear only on some). The lists are also in different languages, and drugs often go by different names.
The team has now created a master file of 2,200 medicines, most of them available in Canada. These include common prescription drugs for high blood pressure, asthma, bacterial infections, skin conditions, depression, anxiety, HIV/AIDS, heart disease, diabetes and many other conditions. The researchers are now systematically searching the literature on each drug and analyzing the studies that led to their being approved. The goal: to verify that there is substantial evidence supporting the efficacy of each drug. “When you even just scratch the surface, you realize there are commonly used medications that are not backed by enough evidence,” says Persaud.
He wants to make clear he’s not anti-medication; he’s pro effective medicines. “The point of the essential medicines movement is to weed out ineffective medicines in order to promote access to the most effective treatments.” Drugs that don’t work come with risks and costs: they prevent patients from receiving appropriate treatment, they may have unwanted side-effects and they waste a country’s limited resources.
So how did ineffective medicines get on the market in the first place? Many, including Diclectin, received approval decades ago – before regulators such as Health Canada required clinical trials. Some of these medicines might not be approved today. In other cases, several drugs might act in similar ways, so some could be redundant. As it stands now, there are about 13,000 pharmaceutical products approved for sale in Canada – an unwieldy number for doctors, pharmacists and patients to navigate. Persaud says a carefully curated short list, containing a few hundred essential medicines that have been proven effective, would lead to less confusion and help ensure patients receive the right ones. It could also allow governments in Canada to collectively purchase a smaller number of medications in larger quantities, resulting in cost savings.
Persaud’s interest in essential medicines was sparked in 2010, when a pregnant patient questioned him about Diclectin’s effectiveness. When he couldn’t find any literature supporting it, he made what he thought was a routine request to Health Canada for data. Health Canada said it couldn’t release what it called confidential business information. That led to Persaud’s seven-year hunt, filled with freedom of information requests, before finally receiving data indicating that the medicine did not meet the drug company’s own standards for effectiveness. Health Canada maintains that the drug is safe and effective, but has announced a plan to make information about the safety and efficacy of drugs more publicly available in future.
Results from Persaud’s essential medicines project are expected in two years.
U of T doctor Mike Evans explains why the surplus of choice in medications is a problem
Recent Posts
People Worry That AI Will Replace Workers. But It Could Make Some More Productive
These scholars say artificial intelligence could help reduce income inequality
A Sentinel for Global Health
AI is promising a better – and faster – way to monitor the world for emerging medical threats
The Age of Deception
AI is generating a disinformation arms race. The window to stop it may be closing